Abstract
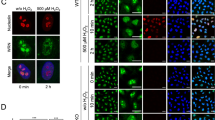
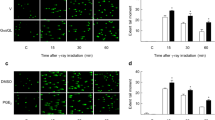
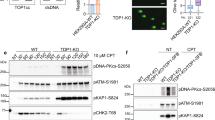
Camptothecin (CPT) and X-ray (XR) generate double-strand breaks (DSB) that can be processed by homologous or nonhomologous recombination. We studied the participation of proteins involved in recombination pathways and cell cycle control in the signal transduction between DNA damage and NF-κB. Cells harbouring mutated NBS, hMRE11, BRCA1 or MLH1 were analysed. NBS- and hMRE11-deficient cells present a classical kinetic of NF-κB induction after camptothecin treatment. When DSB are generated by XR, NBS-deficient cells exhibit a delayed and strongly reduced level of NF-κB induction, whereas the hMRE11 mutated cells do not induce NF-κB at all. This indicates an important role of the hMRE11/hRAD50/NBS complex in the signal transduction initiated by XR. In HCC1937 cells that express a truncated version of BRCA1, XR induces a very rapid and transient NF-κB activation, whereas CPT leads to a delayed activation suggesting that BRCA1 modulates the transduction pathways in different manners after these two stresses. Finally, we found that a proficient MMR pathway is essential to the NF-κB activation after both CPT and XR. These results indicate that DSB originating from XR or CPT do not induce NF-κB in a unique way. MMR participates in both cascades, whereas the hMRE11/hRAD50/NBS trimer is specifically involved in the response elicited by XR.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Avemann K, Knippers R, Koller T and Sogo J . (1998). Mol. Cell. Biol., 8, 3026–3024.
Bharti A and Aggarwal B . (2002). Biochem. Pharmacol., 64, 883–888.
Brown K, Rathi A, Kamath R, Beardsley D, Zhan Q, Mannino J and Baskaran R . (2003). Nat. Genet., 33, 80–84.
Carney J, Maser R, Olivares H, Davis E, Le Beau M, Yates III J, Hayes L, Morgan W and Petrini J . (1998). Cell, 93, 477–486.
Cerosaletti K, Desai-Mehta A, Yeo T, Kraakman-Van Der Zwet M, Zdzienicka M and Concannon P . (2000). Mutagenesis, 15, 281–286.
Cortez D, Wang Y, Qin J and Elledge S . (1999). Science, 286, 1162–1166.
D'Amours D and Jackson S . (2002). Nat. Rev., 3, 317–327.
Friedberg EC, Walker JGC and Siede W . (1995). DNA Repair and Mutagenesis. ASM Press: Washington, DC.
Furuta T, Takemura H, Liao Z-Y, Aune G, Redon C, Sedelnikova O, Pilch D, Rogakou E, Celeste A, Chen H, Nussenzweig A, Aladlem M, Bonner W and Pommier Y . (2003). J. Biol. Chem. 278, 20303–20315.
Gatei M, Young D, Cerosaletti K, Desai-Mehta A, Spring K, Kozlov S, Lavin M, Gatti R, Concannon P and Khanna K . (2000). Nat. Genet., 25, 115–119.
Gosh S and Karin M . (2002). Cell, 109 (Suppl.), S81–S96.
Giannini G, Ristori E, Cerignoli F, Rinaldi C, Zani M, Viel A, Ottini L, Crescenzi M, Martinotti S, Bignami M, Frati L, Screpanti I and Gulino A . (2002). EMBO Rep., 3, 248–254.
Habraken Y, Piret B and Piette J . (2001). Biochem. Pharmacol., 62, 603–616.
Harfe B and Jinks-Robertson S . (2000). Annu. Rev. Genet., 34, 359–399.
Huang T, Wuerzberger-Davis S, Seufzer B, Shumway S, Kurama T, Boothman D and Miyamoto S . (2000). J. Biol. Chem., 275, 9501–9509.
Hur G, Lewis J, Yang Q, Lin Y, Nakano H, Nedospasov S and Liu Z . (2003). Gene Dev., 17, 873–882.
Jacob S and Praz F . (2002). Biochimie, 84, 27–47.
Jacob S, Aguado M, Fallik D and Praz F . (2001). Cancer Res., 61, 6555–6562.
Karin M, Cao Y, Greten F and Li Z . (2002). Nat. Rev. Cancer, 2, 301–310.
Koi M, Umar A, Chauhan D, Cherian S, Carethers J, Kunkel T and Boland C . (1994). Cancer Res., 54, 4308–4312.
Kraakman-Van Der Zwet M, Overkamp W, Friedl A, Klein B, Verhaegh G, Jaspers N, Midro A, Eckardt-Schupp F, Lohman P and Zdzienicka M . (1999). Mutat. Res., 434, 127.
Li N, Banin S, Ouyang H, Li G, Courtois G, Shiloh Y, Karin M and Rotman G . (2001). J. Biol. Chem., 276, 8898–8903.
Lim D-S, Kim T, Xu B, Maser R, Lin J, Petrini J and Kastan M . (2000). Nature, 404, 613–617.
Liu C, Pouliot J and Nash H . (2002). Proc. Natl. Acad. Sci., 99, 14970–14975.
Mirzeova O and Petrini J . (2001). Mol. Cell. Biol., 21, 281–288.
Paull T, Cortez D, Bowers B, Elledge S and Gellert M . (2001). Proc. Natl. Acad. Sci., 98, 6086–6091.
Petrini J . (2000). Curr. Opin. Cell Biol., 12, 293–296.
Pichierri P, Franchitto A, Piergentili R, Colussi C and Palitti F . (2001). Carcinogenesis, 22, 1781–1787.
Piret B and Piette J . (1996). Nucleic Acid Res., 24, 4242–4248.
Piret B, Schoonbroodt S and Piette J . (1999). Oncogene, 18, 2261–2271.
Pommier Y, Pourquier P, Fan Y and Strumberg D . (1998). Biochim. Biophys. Acta, 1400, 83–105.
Shiloh Y . (1997). Ann. Rev. Genet., 31, 635–662.
Scully R, Ganesan S, Vlasakova K, Chen J, Socolovsky M and Livingston D . (1999). Mol. Cell, 4, 1093–1097.
Stewart G, Maser R, Stankovic T, Bressan D, Kaplan M, Jaspers N, Raams A, Byrd P, Petrini J and Taylor A . (1999). Cell, 99, 577–587.
Thompson L and Schild D . (2002). Mut. Res., 509, 49–78.
Vance J and Wison T . (2002). Proc. Natl. Acad. Sci., 99, 13669–13674.
Varon R, Vissinga C, Platzer M, Cerosaletti K, Chrzanowska K, Saar K, Beckmann G, Seemanova E, Cooper P, Nowak N, Stumm M, Weemaes C, Gatti R, Wilson R, Digweed M, Rosenthal A, Sperling K, Concannon P and Reis A . (1998). Cell, 93, 467–476.
Venkitaraman A . (2001). J. Cell Sci., 114, 3591–3598.
Xu B, Kim S, Lim D and Kastan M . (2002). Mol. Cell. Biol., 22, 1049–1059.
Wu X, Petrini J, Heinse W, Weaver D, Livingston D and Chen J . (2000a). Science, 289, 11a.
Wu X, Ranganathan V, Weisman D, Heine W, Ciccone D, O'Neill T, Crick K, Pierce K, Lane W, Rathbun G, Livingston D and Weaver D . (2000b). Nature, 405, 477–482.
Zhao S, Weng Y, Yuan S, Lin Y, Hsu H, Lin S, Gerbino E, Song M, Zdzienicka M, Gatti R, Shay J, Ziv Y, Shiloh Y and Lee E . (2000). Nature, 405, 473–477.
Zhong Q, Boyer T, Chen P and Lee W . (2002). Cancer Res., 62, 3966–3970.
Zhong Q, Chen C, Li S, Chen Y, Wang C-C, Xiao J, Chen P-L, Sharp Z and Lee W . (1999). Science, 285, 747–750.
Acknowledgements
We are indebted to Dr M Zdzienicka (Leiden University, Netherlands) who kindly supplied us NBS-ILB1 cells and to Dr P Concannon (Virginia Mason Research Center, Seattle, USA) who sent us NBS-ILB1-6 cells. CWAT, ATLD2 and ATLD3 cells were a generous gift from Dr M Taylor (University of Birmingham, CRC Institute for Cancer Research, Birmingham, UK). We also thank Dr R Boland (UCSF, La Jolla, USA) for allowing us to use his corrected HCT116 3-6 cells. We thank Ch De Jesus for his assistance in this work. This work was supported by grants from the Belgian National Fund for Scientific Research (NFSR, Brussels, Belgium), from an IAP program (P5/12) and from a starting grant from the University of Liège. YH is a Research Scientist and JP is a Research Director, both supported by the NFSR. OJ is an assistant from the University of Liège.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Habraken, Y., Jolois, O. & Piette, J. Differential involvement of the hMRE11/hRAD50/NBS1 complex, BRCA1 and MLH1 in NF-κB activation by camptothecin and X-ray. Oncogene 22, 6090–6099 (2003). https://doi.org/10.1038/sj.onc.1206893
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1206893
Keywords
This article is cited by
-
Alteration in stemness causes exclusivity between Epstein–Barr virus-positivity and microsatellite instability status in gastric cancer
Gastric Cancer (2021)
-
DNA damage response signaling pathways and targets for radiotherapy sensitization in cancer
Signal Transduction and Targeted Therapy (2020)
-
NF-κB is a critical mediator of BRCA1-induced chemoresistance
Oncogene (2014)