Article Text
Abstract
A case study of a 38-year-old woman with a diagnosis of placental site trophoblastic tumor is presented. The patient had a 22-month history of amenorrhea since her last pregnancy, and a dilation and curettage procedure was performed after a 3.1×2.4×2.8 cm endometrial echogenic lesion was visualized on a pelvic ultrasound. When the diagnosis of placental site trophoblastic tumor was made by histopathologic and immunohistochemical analysis, complementary examinations including including pelvic magnetic resonance imaging (MRI) and a chest computed tomography (CT) were done. There was no evidence of disease outside the uterus, and a laparoscopic hysterectomy with bilateral salpingectomy was performed. After a surveillance period of 12 months, no disease recurrence was identified. Best imaging studies, treatment options, and proper surveillance for these type of tumors are discussed alongside the case study.
- placental site trophoblastic tumor
- gestational trophoblastic neoplasia
Statistics from Altmetric.com
Case presentation
A 38-year-old woman G2P2 presented to an outside institution with a 22-month history of amenorrhea since her last pregnancy. She had been taking oral contraceptives (estradiol valerate and dienogest) for 15 months. Prior to her pregnancy, the patient had a history of regular menstrual cycles and normal Pap smears. The patient did not have a significant medical history and surgical history was significant only for a laparotomy for a hemorrhagic ovarian cyst. Family history was unremarkable. She had had two term pregnancies, both with normal vaginal deliveries. Although she was taking oral contraceptives, a pelvic ultrasound was performed due to the amenorrhea.
Dr Chacon
Please describe in detailthe findings on the pelvicultrasound
A retroverted uterus of7.6×5.7×4.2 cmwith a3.1×2.4×2.8 cmendometrial echogenic lesion was noted. It was described as a hypervascularized image with low-resistance blood flow, and interpreted as a probable uterine polyp. No atypical findings were described on the ovaries. The right ovary measured3.2×2.9 cmand the left one3.4×3.1 cm. Both had at least five follicle-like images of less than0.6 cm.
The patient was taken to the operating room for dilation and curettage. A hysteroscopy was not performed because this resource was not available in that institution. Histopathologic examination of the extracted tissue was compatible with placental site trophoblastic tumor. After that, she was referred to our hospital.
On initial presentation to our institution, physical examination showed a soft, nontender, and nondistended abdomen. On pelvic examination, neither the uterus nor the ovaries were enlarged. Laboratory studies were all normal except for a serum beta human chorionic gonadotropin (b-hCG) that was 6 mU/mL (normal value <5.3 mU/mL). Serum human placental lactogen was not measured, as it was not considered useful in monitoring the course of this disease or guiding clinical management. Histological and immunohistochemical analysis of the original biopsy was performed and the diagnosis of placental site trophoblastic tumor was confirmed. A pelvic MRI was then performed.
Dr Wernicke
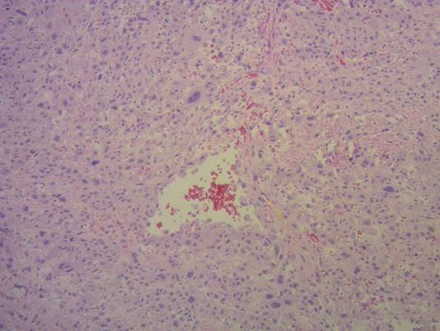
On microscopic analysis,thetumor was composed of polygonal implantation site intermediate trophoblastic cells with irregular, hyperchromatic nuclei, and dense eosinophilic to amphophilic cytoplasm with occasional vacuoles. The implantation site intermediate trophoblastic cells were characteristically separating muscle bundles as they invaded the myometrium. Hemorrhagic areas were observed. (Figure 1)
Microscopy showing mononuclear intermediate trophoblastic cells, increased mitotic activity, and destructive invasion of myometrium (A) hematoxylin and eosin (H&E)×2.5 (B) H&E×4.
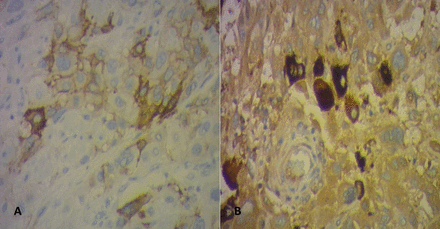
Immunohistochemical stains showed tumor cells strongly and diffusely positive for human placental lactogen. Placental alkaline phosphatase andhuman chorionic gonadotropin(hCG) immunostain showed focal positivity(Figure 2), and Ki-67 proliferation index was20%to35%.
Immunohistochemical testing showing focal positivity for placental alkaline phosphatase (A) and hCG (B) ×100.
Dr Xiang
Given the diagnosis of placental site trophoblastic tumor, what is considered the best imaging modality for this patient? Should imaging of the brain also be routinely included in patients with this diagnosis?
Placental-site trophoblastic tumor is a rare type of gestational trophoblastic neoplasia. Different from invasive mole and choriocarcinoma, which can be clinically diagnosed,placental-site trophoblastic tumor requires pathological diagnosis, and has specific pathological and immunohistochemical characteristics. It is important to assess the extent and the metastatic spread of disease thoroughly before starting treatment after pathological confirmation. There are two main views on imaging evaluation. In the United States, initial evaluation with abdomen-pelvis MRI, chest CT, and brain MRI is recommended.1FIGO/EOTTD recommends that only pelvic-transvaginal Doppler ultrasound and chest X-ray should be initially requested inpatientswith post-molar gestational trophoblastic neoplasia. Only if the X-ray is inconclusive or the lesion is greater than1 cm, should chest CT and brain MRI be complemented.
Considering the different perspectives, image evaluation for placental-site trophoblastic tumor in this patient include the flowing two aspects:
1 Evaluation ofprimarytumor.Pelvic ultrasound examination is actually a rough assessment,and isnot specific. At present, the gray-scale and DopplerUltrasoundappearance of placental-site trophoblastic tumor is not specific and sometimesitmay be difficult to differentiate placental-site trophoblastic tumor from benign uterine lesions (eg,fibroids, adenomyomas).2So, when assessing the extent of disease, it may be confused with concomitant benign diseases. MRI can show the location, size, and depth of infiltration of the lesion on the uterus. It can help to find whether it is a sharply marginated or ill-defined mass centered within the myometrium that distorts or displaces the junctional zone. In MRI,3placental-site trophoblastic tumor may present as myometrial masses that are isointense to healthy myometrium on T1-weighted images and isointense to slightly hyperintense on T2-weighted images. In some cases, MRI can accuratelylocalize the placental-site trophoblastic tumor lesion, which was invisible in sonography, and allowpatientsto be treated with hysterotomy rather than with hysterectomy. Therefore, we believe that MRI is more advantageous in the evaluation of uterine lesions.
2 Evaluation ofdistantmetastases.The most common site of metastasis is the lung, with an incidence at diagnosis of10%to29%. Less common sites of metastasis are represented by the liver, brain, spinal cord, ovary, vagina, peritoneum, spleen, and pancreas.4The prevalence of brain metastases in gestational trophoblastic neoplasia. ranges from3.4%to8.8%,5 6but the incidence in placental-site trophoblastic tumor was not reported. A previous multi-center data showed that among 108 placental-site trophoblastic tumor cases,7only twopatientshad stageIV, with an incidence of brain metastasis less than2%. In general,patientswith lung metastases are at increased risk for central nervous system metastases. Considering the pathophysiological basis of hematogenousmetastasis, placental-site trophoblastic tumorpatientswith no evidence of lung metastasis are not required to have brain MRI routinely.
Dr Chacon
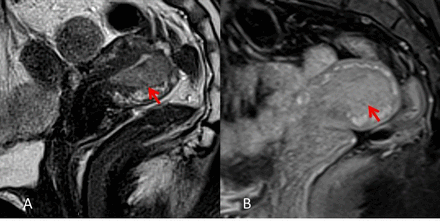
Pelvic MRI (Figures 3 and 4) showed a3.8×3.5×1.8 cmsolid lesion in the posterior endometrial wall infiltrating the internal myometrium and sparing the external myometrium, except for probable deep infiltration in the fundus. The uterine serosa and the cervix were spared. The lesion had intermediate signal on T2, signal restriction on diffusion-weighted imaging, and apparent coefficient measurement (DWI/ADC), as well as early and avid enhancement on dynamic sequences. Both ovaries were normal and a3 cmfunctional cyst was seen on the left ovary. No free fluid, adenopathy, or suspicious bone lesions were detected. Abdominal MRI and chestCTdid not show evidence of distant disease.
Pelvic MRI. Sagittal T2 WI (A) and Sagittal T1 gadolinium WI (B) showing a 3.8x.3.5x1.8 cm lesion in the posterior uterine wall. WI, weighted imaging.
Pelvic MRI (axial sequences) showing a solid lesion with intermediate signal on T2 (A), early and avid enhacement on dynamic gadolinium sequences (B), and signal restriction on DWI ADC (C,D). DWI, diffusion weighted imaging; ADC, apparent diffusion coefficient
Dr Xiang
What options of therapy should be discussed and offered to the patient? Is there any role for preoperative chemotherapy in a patient with this diagnosis? Please provide information regarding the role of oophorectomy in a patient with this diagnosis.
At present, thepatient’shCGis slightly elevated, andthepathology report demonstratesplacental-site trophoblastic tumor. Thepatientwas confirmed as FIGO stage1by a comprehensive assessment of the lesion extent. Treatment options for thispatientincludehysterectomy.Total hysterectomy is a choice for placental-site trophoblastic tumorpatientswith early-stage, because the disease is less sensitiveto chemotherapy. For thispatient, MRI showed a clear-margined lesion within the muscular layer and sparing the external myometrium. Abdominal MRI and pulmonary CT did not indicate any metastasis. A large portion ofpatients, whoselesion is confinedto the uterus, achieve complete remission with surgery alone.Another option could be aconservative management.Fertility-sparing treatment is feasible for youngpatientswith placental-site trophoblastic tumor apparently confined to the uterine body but is not suitable in diffuse lesions. The treatment includes transabdominal focal uterine resection or hysteroscopic (or combined with laparoscopic) hysterotomy, depending on lesion size and its relationship with the muscular layer. When discussing the treatment plan, it is necessary to know whether thepatienthas the desire to retain the fertility and thepatientrequirescareful counseling.
Placental-site trophoblastic tumor is far less sensitive to chemotherapy than other types of gestational trophoblastic neoplasia such as invasive mole and choriocarcinoma. Surgery is the primary mode of treatment in most cases and plays an important role in metastatic disease. Surgery combined with chemotherapy was commonly recommended inpatientswith stagesII–IV.Preoperative chemotherapy has not been proven to benefit placental-site trophoblastic tumorpatients.
Ovarian metastases are uncommon and oophorectomy does not seem to improve survival If no adnexal metastasis is found during the operation.
After counseling, the patient expressed no desire for fertility preservation, thus a hysterectomy was proposed as treatment. She was operated by the laparoscopic approach in August 2018. Intraoperative findings showed engorged pelvic vessels, but no other abnormalites. The uterus measured approximately 8 cm x 4 cm. A hysterectomy with bilateral salpingectomy was performed. Estimated blood loss was less than 100 mL, and the surgical time was 120 min. There were no intraoperative complications. The patient was discharged on postoperative day one.
Dr Wernicke
The gross tissue evaluation showed an exophytic well-circumscribedtumorousformation in the posterior uterine wall, of yellowish-white coloration and firm consistency measuring approximately3.2×2.0×1.8 cm. Focal hemorrihage and necrosis was also noted.(figure 5) On microscopic analysis, the tumor invaded the myometriumby15 mm, being located4 mmfrom the uterine serosa. Large confluent sheets of mononuclear, polygonal implantation site intermediate trophoblastic cells were seen, and perivascular invasion was evinced. (figure 6)
Well-circumscribed lesion in the posterior uterine wall.
Neoplastic cells were surrounding and invading a blood vessel, extending into the vascular lumen. The vessel wall is replaced by fibrin.
Dr Xiang
In this setting, would there be a role for adjuvant chemotherapy or radiotherapy?
What would be the proposed surveillance for such a patient?
The preoperativehCGlevel was slightly elevated, and MRI imaging showed a clear-margin lesion smaller than4 cm. The gross pathology presented as a exophytic well-circumscribedtumorousformation in the posterior uterine wall, measuringapproximately3.2×2.0×1.8 cm. On microscopic analysis, focal hemorrhage and necrosis werenoted and perivascular invasion was evinced. From the above description, there were no high-risk factors for poor prognoses.
Forpatientswith low-risk stage I disease, there is no indication for adjuvant chemotherapy after hysterectomy. Adjuvant chemotherapy is usually suggested8forpatientswith either an interval from the antecedent pregnancy >4yearsor those with deep myometrial invasion or serosal involvement or high mitotic index or for those with stagesII–IV disease. Individualized radiotherapy can be sometimes considered. For this case, as there were no high-risk factors, if β-hCG level decreased satisfactory after surgery, adjuvant chemotherapy was not recommended, neither was radiotherapy.
β-hCG levels should be monitored regularly.Patientsshould be monitored every month for the first3 monthsafter the end of the treatment. Subsequently, the serum β-hCG levels are monitored every3monthsfor9months, followed by every6monthsfor2years. Pelvic MRI or chest CT should be performedif necessary.
The patient has been followed with history and physical examination every 6 months, and a β-hCG measurement with a CT scan at 12 months from surgery. On last evaluation in August 2019, the patient had no evidence of disease. Hystory and physical examination were unremarkable, β-hCG was <2.0 mU/mL, and a CT of the chest, abdomen, and pelvis showed no lesions.
Dr Xiang
Closing summary
Placental-site trophoblastic tumor is a rare form of gestational trophoblastic neoplasia, arising from intermediate trophoblast of the implantation site. The presenting symptom of placental-site trophoblastic tumor is usually untypical, and serumβ-hCGlevels are usually only modestly elevated or normal in placental-site trophoblastic tumor. Accurate diagnosis depends on histopathology with special immunohistochemical staining.
Imaging plays an important role in assessing the local extent of disease and systemic surveillance. Ultrasound is a preliminary examination but not specific. The MRI appearance of placental-site trophoblastic tumor is not specific as well, but MRI is superior toultrasound in evaluating tumor location and extension of extrauterine pelvic tumor. MRI may also be useful in tumors that are poorly visualized atultrasound. The significance of MRI application in placental-site trophoblastic tumor is to provide the basis for conservative treatment.The most common site of metastasis is the lungs, thereforeimaging of the lungs is usually recommended. Chest CT is much more sensitive and allows detection of lung metastases with greater accuracy. MRI imaging of the brain and CT of the abdomen are recommended to those with lung metastases, and such findings may substantially alter staging and subsequent management.
Surgery is the most appropriate treatment option in most cases of placental-site trophoblastic tumor, since itis less chemosensitive than invasive moleor choriocarcinoma. Simple hysterectomy is the preferred treatment for mostpatients. Stage Ipatientscan achieve complete remission with surgery alone. Adjuvant chemotherapydoes not seem to offer any significant benefit in this clinical setting.Patientswith extensive metastasis or recurrence may receive adjuvant chemotherapy. In the past15years, Peking Union Medical College hospital has received a total of 57 cases of this rare disease, among which the complete response rate of stagesIII–IVpatientscan reach87.5%after combined surgery and chemotherapy.However,randomizedtrials have not been performed because of the rarity of this tumor. Adjuvant chemotherapy is usually suggested in the following situation:metastatic lesions outside the uterus;FIGO stage I but with other adverse prognostic factors, such aslong intervalfromthe antecedent pregnancy (>4years), vascular infiltration, deep myometrial invasion, andhigh mitotic index;and if serum β-HCG level continues to rise after surgery.
Footnotes
Twitter @guidoreyv
Funding The authors have not declared a specific grant for this research from any funding agency in the public, commercial, or not-for-profit sectors.
Competing interests None declared.
Patient consent for publication Obtained.
Provenance and peer review Commissioned; internally peer reviewed.